|
5/28/2023 0 Comments Dmg reaction with nickel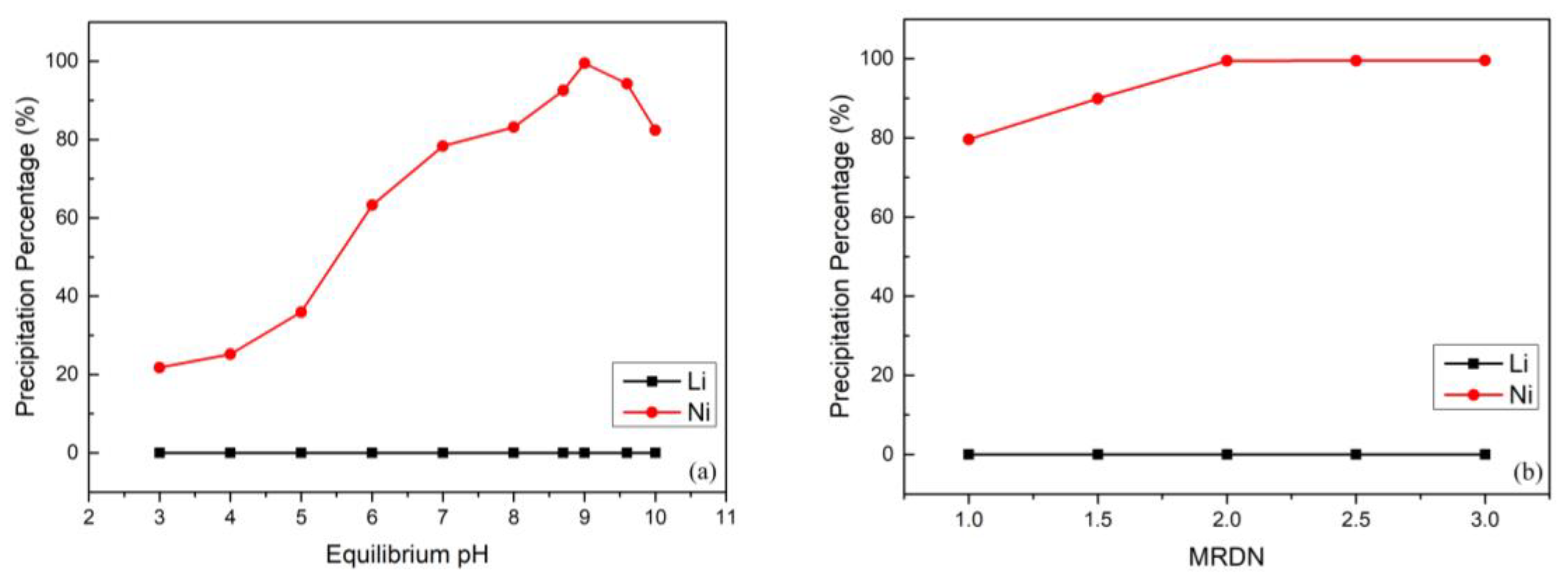
Before the five-cent piece can be analyzed for the nickel content, it must be cleaned, weighed and then dissolved. The Ni(DMG) 2 complex (MW 288.9 g/mole) is stable to heat and is unusually hydrophobic it will dry quickly in an oven at 110-120 oC to yield a pure substance whose weight can be used to calculate the nickel content of the original sample.Ī five-cent piece (a nickel) consists of copper alloyed with nickel and other metals. The reaction is illustrated below the dashed lines represent hydrogen bonds which help stabilize the complex.Ģ Dimethylglyoxime + Ni 2+ -> Ni(DMG) 2 + 2 H + No other common metallic ion reacts in the same way, so the formation of the insoluble Ni(DMG) 2 complex makes it possible to separate the Ni 2+ ions from other metal ions in solution. In the case of nickel, Ni 2+ ions form an extremely insoluble bright red complex with two dimethylglyoxime (DMG) anions under mildly basic conditions. That usually means that a material dissolved in solution is completely precipitated from solution, and the solid is weighed. 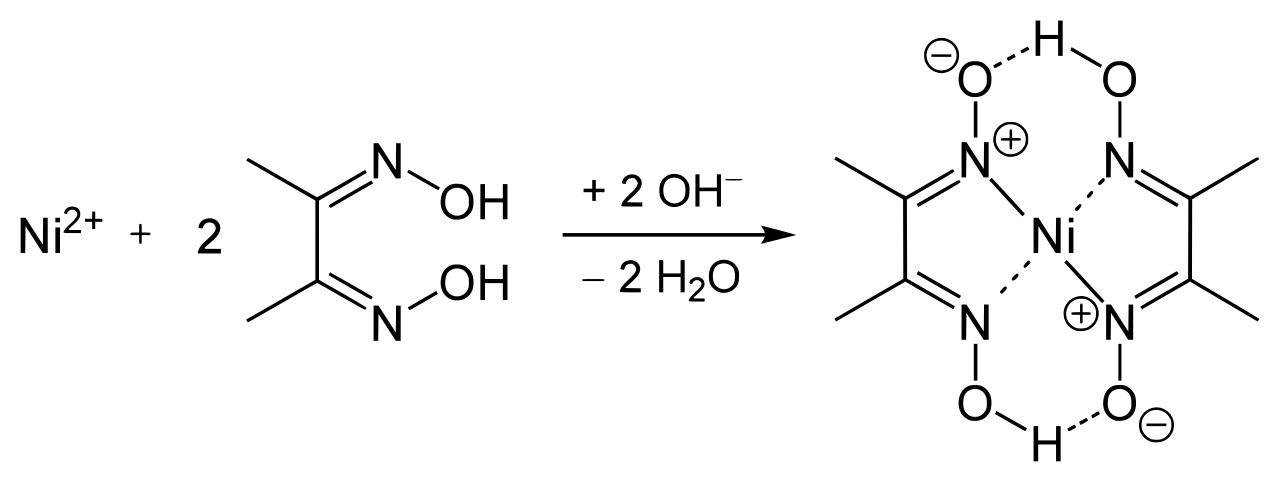 
Experiment 7-023: Gravimetric determination of Nickel in a Five-cent CoinĪ gravimetric determination of a substance involves the separation of that substance in a form which may be weighed accurately and is of known composition.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
